Ethical Conundrums of Disclosing Dementia Research Results with Participants
GBHI faculty member Lorina Naci shares challenges and guidance on the ethical sharing of dementia research results.
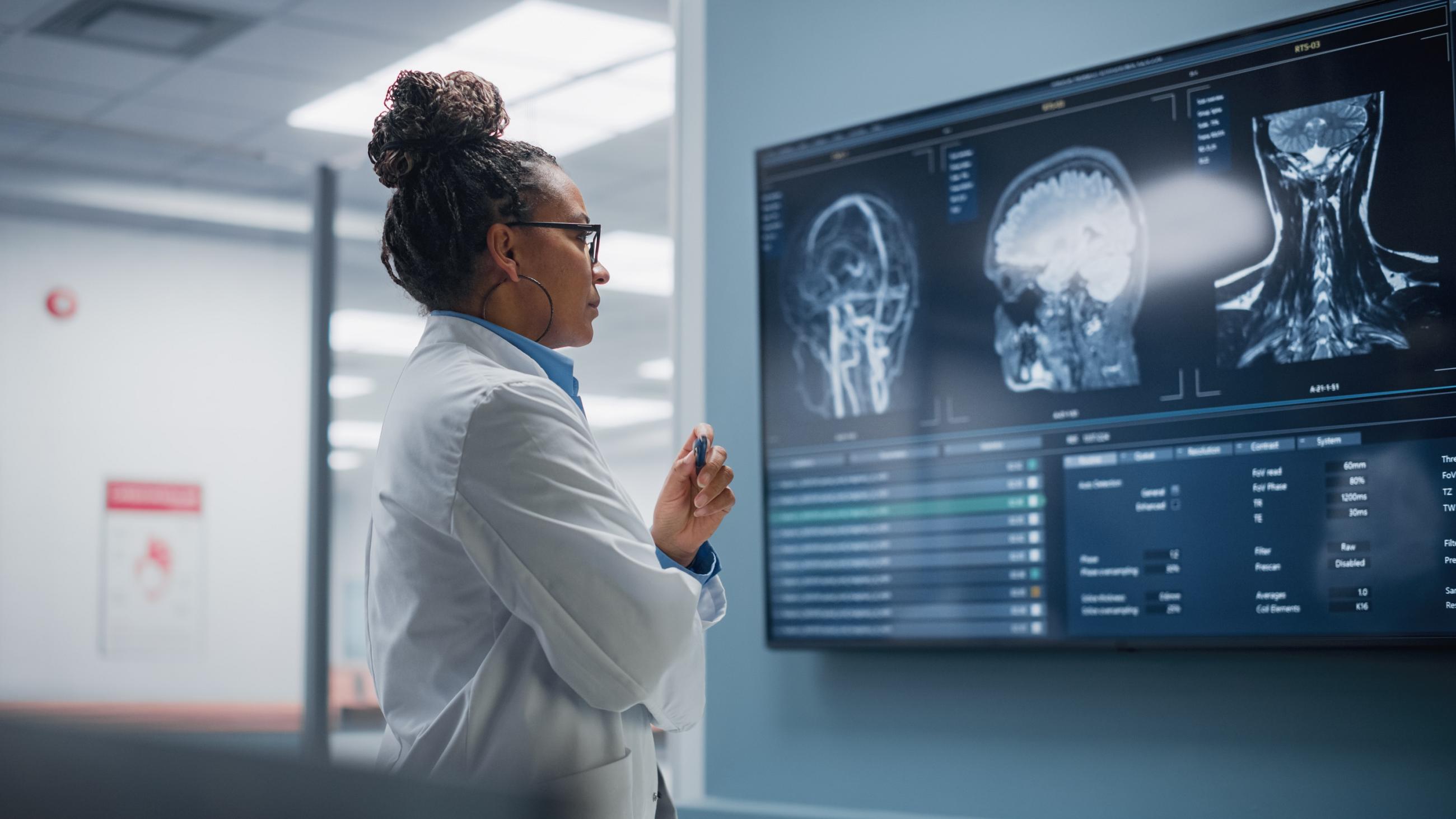
Dementia is a global pandemic that presents profound challenges to health care systems, families, and societies throughout the world. Growing research efforts aim to identify individuals at greatest risk of developing late-onset sporadic Alzheimer’s disease early, in order to deliver targeted risk reduction and prevention interventions from midlife. However, current practice guidelines recommend against disclosing individual-level dementia risk to cognitively unimpaired individuals due to low predictive value (Dubois et al. 2021) and ethical challenges. Given increasing understanding of individual choice in the promotion of brain health, what good can brain health research do if we can’t share research results with individual participants?
The principles of respect for autonomy, doing good/doing no harm, and justice require a nuanced interpretation in the context of fear of dementia. To explore these issues, I recently organized a roundtable with an international panel of experts including members of the Global Brain Health Institute (GBHI) community
Compounding Effect of Fear of Dementia
Fear of dementia studies have shown that for a significant portion of the population (26–39%), dementia is the most feared medical diagnosis, surpassing even cancer. Stark recent findings show an increased risk of suicide attempt in individuals who received a recent diagnosis of mild cognitive impairment or dementia (Günak et al. 2021). Furthermore, concern about developing dementia, combined with a general lack of knowledge about Alzheimer’s disease and other types of dementia, has been argued to contribute to “dementia worry, which is associated with poorer cognitive functioning” and can have a compounding effect, by leading to avoidance of behaviors that protect against dementia, such as engaging in socially and intellectually stimulating activities. This association between fear of dementia and negative mental, social, and physical outcomes complicates the question of whether sharing aggregate research findings is beneficial or harmful to participants.
Recommendations on the Ethical Sharing of Dementia Research Results
Only about one-third of research participants receive any results from studies they have participated in. This represents a potential missed opportunity to acknowledge and recognize the important contribution that participants have made to research, potentially satisfy their curiosity about the outcomes of the research, and to possibly engage participants in further research.
In a recent paper published in the Cambridge Quarterly of Healthcare Ethics, we provide the following guidelines on how to navigate this complicated territory:
- If the decision is made to offer results, it is important that the research protocol includes specific information about what information will be shared, so that it can be reviewed both by the Research Ethics Committee and by participant representatives or community partners.
- It is important that the consent form indicates what, when, how, and with whom data will be disseminated, and provide sufficient detail for participants to make an informed choice about receiving results, as well as the option to participate in the study without receiving aggregate results.
- It may be necessary for participants to go through a separate informed consent process at the conclusion of the study, prior to receiving aggregate results, to ensure that the participant has not changed their mind about receiving this information, and to give researchers an opportunity to inform them about new or unexpected information that was not conveyed during the initial informed consent process.
- A clear strategy for communicating results should also be included as part of the research protocol, including what support will be provided to participants receiving research results.
Developing a communication strategy may require engaging with representatives of the research population, to identify their informational needs and preferences. For example, research suggests that participants prefer face-to-face communication when receiving feedback about research results, especially when findings relate to severe or untreatable conditions.
In summary, communicating risk factors for future dementia with research participants is a timely and important issue that requires careful consideration from researchers and medical professionals in consultation with the general public. Recent research also shows that 9 out of 10 people want to know their risk of brain disease (Carver et al., 2022). With the exponential increase in focus and research on brain health—as a holistic state and set of processes that can be actively promoted through lifestyle choices—ethical guidelines for communicating risk factors to cognitively healthy individuals are urgently needed.
Reference:
Graham M. Farina F, Ritchie C, Lawlor B, Naci L. (2022) Fear of dementia and public sharing of research results. Cambridge Quarterly of Healthcare Ethics. 31: 4, 498–505
Authors

Lorina Naci, PhD
Professor in Psychology
GBHI Members Mentioned

Francesca Farina, PhD
Neuroscientist

Brian Lawlor, MD, FRCPI, FRCPsych, MRIA
Founding Director, Trinity College Dublin


